Research Interests
Research Background
Lyophilization, better known as freeze drying, is an increasingly common step in development and manufacture of pharmaceuticals—in the last two decades, annual FDA approvals of new lyophilized drug products have increased threefold. Many biologically-derived treatments have a naturally short lifetime in the aqueous state. In a lyophilized solid state, their shelf life can be months or years without refrigeration, in contrast to the ultracold conditions used to ship and store COVID-19 mRNA vaccines. However, lyophilization is a batch process that takes as much as two weeks, hampering both cost and throughput. Process models for lyophilization mitigate this tradeoff by maximizing drying rate within temperature limits.
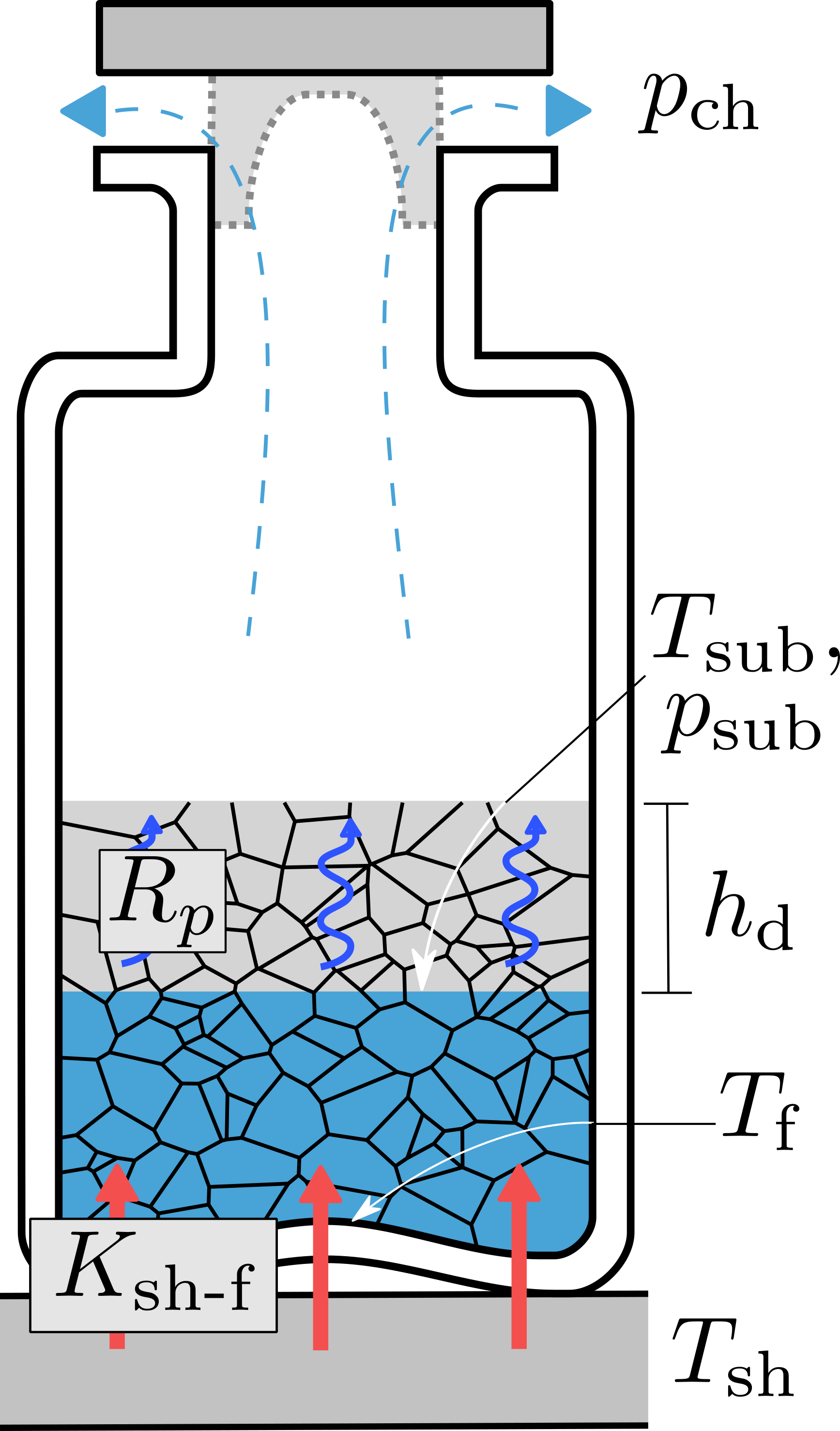
My experience, both at Purdue and during my Fulbright grant at the Politecnico di Torino in Italy, is in developing and refining new physics-based models, building on the (40-year-old) state-of-the-art model for temperature in a vial during lyophilization. Though my passion lies especially in theory and computational research, models are most useful when they predict reality, so I have gathered, interpreted, and analyzed experimental data to assess model validity. This is true for both my work on experimentally-backed models for microwave heating and rarefied flow in lyophilized porous structures. With that background, I am equipped to bring together experiment, theory, and simulation for the “AI-enabled pharmaceutical innovation” called for by a recent accord on US pharmaceutical manufacturing.
Proposed Future Directions
Emphasis 1: open source models with novice-friendly interfaces
 Experience (including mine) shows that code is essentially useless without good documentation and tutorials. Similarly, new science is most useful to industry when it does not take an expert to digest. My experience maintaining an industrially-relevant Python library for lyophilization process design confirms that this does not happen automatically. Models need to have a simple, accessible interface (like a web GUI or mobile app) to see adoption outside expert communities, and these interfaces need ongoing maintenance. By creating and maintaining these interfaces, my research group will make models (both the equations and the code) accessible to industry, as well as to colleagues in other academic units. In contrast to previous efforts in the field of lyophilization, we will draw on software engineering best practices (like unit testing and continuous integration) to ensure models and their interfaces are reliable and maintainable. A first case will be developing an interactive application for microwave-assisted lyophilization, then using it to develop and showcase a new design space analogous to what is used for conventional lyophilization. Test suites ensure model implementations remain reliable and maintainable, justifying their use in the regulatory approval processes which shape the pharmaceutical industry.
Experience (including mine) shows that code is essentially useless without good documentation and tutorials. Similarly, new science is most useful to industry when it does not take an expert to digest. My experience maintaining an industrially-relevant Python library for lyophilization process design confirms that this does not happen automatically. Models need to have a simple, accessible interface (like a web GUI or mobile app) to see adoption outside expert communities, and these interfaces need ongoing maintenance. By creating and maintaining these interfaces, my research group will make models (both the equations and the code) accessible to industry, as well as to colleagues in other academic units. In contrast to previous efforts in the field of lyophilization, we will draw on software engineering best practices (like unit testing and continuous integration) to ensure models and their interfaces are reliable and maintainable. A first case will be developing an interactive application for microwave-assisted lyophilization, then using it to develop and showcase a new design space analogous to what is used for conventional lyophilization. Test suites ensure model implementations remain reliable and maintainable, justifying their use in the regulatory approval processes which shape the pharmaceutical industry.
Emphasis 2: model commonalities across drying technologies
 As highlighted by the Lyo2040 technology roadmap, there are a variety of emerging drying technologies which address the weaknesses of traditional lyophilization, such as spray freeze drying, microwave-assisted lyophilization, continuous spin-freeze-drying, suspended-vial continuous lyophilization, or lyophilization of discrete “lyo beads” or “lyospheres” (typically larger than the droplets in spray freeze drying). The key heat and mass transfer modes differ somewhat from one process to another, and each has its own unique phenomena (such as spray atomization or microwave electromagnetism), but commonalities such as phase equilibrium between ice and vapor will mean lessons learned for one drying approach can be useful to development and regulatory approval for other processes. My lab will work towards developing unified modeling frameworks for understanding these processes, not only in mathematical notation but numerical implementation. As this comparison yields model insights, we will measure experimental process data (e.g. for lyophilization or spray drying) to validate model improvements. For processes where only prototype equipment exists, we will lean on literature data. We will also work to cross-pollinate the insight that can be gained from material-efficient measurements like freeze-drying microscopy, direct scanning calorimetry, or single-drop drying, which already aid process design but are only applied for some of these drying processes. A specific first case for my students to investigate will be to estimate thermodynamic limits and rates of the secondary drying step of lyophilization, drawing on single-drop drying and spray drying experiments.
As highlighted by the Lyo2040 technology roadmap, there are a variety of emerging drying technologies which address the weaknesses of traditional lyophilization, such as spray freeze drying, microwave-assisted lyophilization, continuous spin-freeze-drying, suspended-vial continuous lyophilization, or lyophilization of discrete “lyo beads” or “lyospheres” (typically larger than the droplets in spray freeze drying). The key heat and mass transfer modes differ somewhat from one process to another, and each has its own unique phenomena (such as spray atomization or microwave electromagnetism), but commonalities such as phase equilibrium between ice and vapor will mean lessons learned for one drying approach can be useful to development and regulatory approval for other processes. My lab will work towards developing unified modeling frameworks for understanding these processes, not only in mathematical notation but numerical implementation. As this comparison yields model insights, we will measure experimental process data (e.g. for lyophilization or spray drying) to validate model improvements. For processes where only prototype equipment exists, we will lean on literature data. We will also work to cross-pollinate the insight that can be gained from material-efficient measurements like freeze-drying microscopy, direct scanning calorimetry, or single-drop drying, which already aid process design but are only applied for some of these drying processes. A specific first case for my students to investigate will be to estimate thermodynamic limits and rates of the secondary drying step of lyophilization, drawing on single-drop drying and spray drying experiments.
Emphasis 3: statistical model assessment
Some authors have explored probabilistic descriptions of process inhomogeneity in conventional lyophilization. For example, the well-known “edge effect” of higher-temperature vials at the edge of a shelf can be quantified with a distribution of heat transfer coefficients, or rational safety margins for product temperature can be estimated by linear propagation-of-error, or a batch of simulations with Monte Carlo-sampled parameters can check parameter sensitivity. However, other statistical aspects have gone unexamined, like the uncertainty resulting from ad-hoc fit of mass transfer resistance coefficients to experimental data. A new Bayesian statistical treatment could readily build on these learned lessons and conventional wisdom as priors, while also accounting for uncertainties from experiment and nonlinear aspects of the model and fitting process, and so provide more rigorous safety margins for industry professionals designing lyophilization processes. A Bayesian framework can also naturally incorporate measurements from either industry-standard equipment, like a thermocouple-equipped lyophilizer, or newer measurements like IR thermography, and my students will explore both experimentally. By providing this analysis natively as part of a graphical interface to process models, my group can make modern statistical techniques accessible to both engineers and regulators.
Long-term Direction: porous structure and flow prediction
The drying rate and duration of conventional lyophilization cannot be predicted without understanding the mass transfer resistance of the partially-dried product. The standard approach treats this resistance empirically, which is simple, but this means running a full lyophilization batch to re-measure the resistance after any process change, especially during scale-up. As a result, the current best practices for cycle design are often both material- and time-intensive. My work is directed towards alleviating this problem by working at a general solution. This will require interdisciplinary work on questions only recently being answered by the community: what ice crystal sizes result from freezing an aqueous solution? How much do porous structures deform? What can we measure about a porous structure to predict rarefied flow behavior? Over time, my lab will develop the experimental expertise (such as in pore imaging), computational simulations (like porous flow), and collaborations (with experts and industrial partners) necessary to provide a coherent, general model for mass transfer resistance.
